Spatial IsoDepth Calculation¶
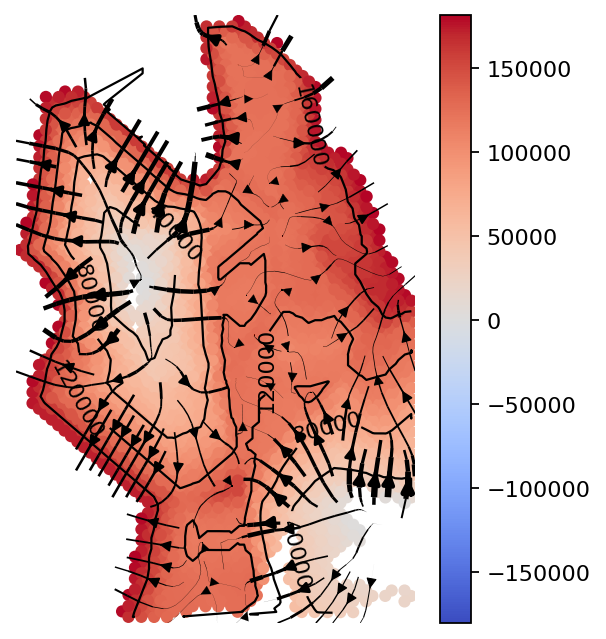
GASTON is an interpretable deep learning model for learning the gene expression topography of a tissue slice from spatially resolved transcriptomics (SRT) data. GASTON models gene expression topography by learning the isodepth, a 1-D coordinate describing continuous gene expression gradients and tissue geometry (i.e. spatial domains).
Here, we introduce method integrated in omicverse named GASTON.
We made three improvements in integrating the GASTON algorithm in OmicVerse:
We reduced the installation conflict of
GASTON, user only need to update OmicVerse to the latest version.We optimized the visualization of
GASTONand simplify the function input ofGASTON.We have fixed some bugs that could occur during function.
Besides, all functions you could find in omicverse.external.GASTON if you want to use the raw functuion to run GASTON’s tutorial
import omicverse as ov
#print(f"omicverse version: {ov.__version__}")
import scanpy as sc
#print(f"scanpy version: {sc.__version__}")
ov.plot_set()
Warning: Could not read dependencies from pyproject.toml: [Errno 2] No such file or directory: '/home/groups/xiaojie/steorra/env/omicverse/lib/python3.10/site-packages/omicverse/pyproject.toml'
____ _ _ __
/ __ \____ ___ (_)___| | / /__ _____________
/ / / / __ `__ \/ / ___/ | / / _ \/ ___/ ___/ _ \
/ /_/ / / / / / / / /__ | |/ / __/ / (__ ) __/
\____/_/ /_/ /_/_/\___/ |___/\___/_/ /____/\___/
Version: 1.6.11, Tutorials: https://omicverse.readthedocs.io/
Prepared stRNA-seq¶
First let’s read spatial Visium data from 10X Space Ranger output. Here we use lymph node data generated by 10X and presented in Kleshchevnikov et al (section 4, Fig 4). This dataset can be conveniently downloaded and imported using scanpy. See this tutorial for a more extensive and practical example of data loading (multiple visium samples).
adata = sc.datasets.visium_sge(sample_id="V1_Human_Lymph_Node")
adata
reading /scratch/users/steorra/analysis/omicverse/omicverse_guide/docs/Tutorials-space/data/V1_Human_Lymph_Node/filtered_feature_bc_matrix.h5
(0:00:01)
AnnData object with n_obs × n_vars = 4035 × 36601
obs: 'in_tissue', 'array_row', 'array_col'
var: 'gene_ids', 'feature_types', 'genome'
uns: 'spatial'
obsm: 'spatial'
adata.var_names_make_unique()
adata.obs_names_make_unique()
Use top PCs of analytic Pearson residuals¶
Here we compute PCA on analytic Pearson residuals following the Scanpy tutorial https://scanpy-tutorials.readthedocs.io/en/latest/tutorial_pearson_residuals.html tutorial . This is faster than GLM-PCA, but the PCs are of lower quality, so it is not recommended
gas_obj=ov.space.GASTON(adata)
gas_obj.get_gaston_input(get_rgb=True,spot_umi_threshold=50)
A=gas_obj.get_top_pearson_residuals(num_dims=8,clip=0.01,n_top_genes=5000,
use_RGB=True)
filtered out 1 cells that have less than 50 counts
Adding image layer `img1`
calculating RGB
Calculating features `['summary']` using `1` core(s)
Adding `adata.obsm['features']`
Finish (0:00:30)
extracting highly variable genes
--> added
'highly_variable', boolean vector (adata.var)
'highly_variable_rank', float vector (adata.var)
'highly_variable_nbatches', int vector (adata.var)
'highly_variable_intersection', boolean vector (adata.var)
'means', float vector (adata.var)
'variances', float vector (adata.var)
'residual_variances', float vector (adata.var)
normalizing counts per cell
finished (0:00:00)
computing analytic Pearson residuals on adata.X
finished (0:00:00)
computing PCA
with n_comps=8
finished (0:00:00)
We first load GLM-PCs and coordinates and z-score normalize.
gas_obj.load_rescale(A)
Training the GASTON model¶
Next we train the neural network, once for each random initialization.
NEURAL NET PARAMETERS (USER CAN CHANGE) architectures are encoded as list, eg [20,20] means two hidden layers of size 20 hidden neurons
isodepth_arch: architecture for isodepth neural network d(x,y) : R^2 -> R
expression_fn_arch: architecture for 1-D expression function h(w) : R -> R^G
num_epochs: number of epochs to train NN (NOTE: it is sometimes beneficial to train longer)
checkpoint: save model after number of epochs = multiple of checkpoint
out_dir: folder to save model runs
optimizer:
num_restarts=20
out_dir='/scratch/users/steorra/tmp/tmp'
gas_obj.train(
isodepth_arch=[20,20],
expression_fn_arch=[20,20],
num_epochs=5000,
checkpoint=500,
out_dir=out_dir,
optimizer="adam",
num_restarts=20
)
Visualization¶
If you use the model trained above, then figures will closely match the manuscript — but not exactly match — due to PyTorch non-determinism in seeding (see https://github.com/pytorch/pytorch/issues/7068 ).
We also include the model used in the paper for reproducibility.
out_dir='/scratch/users/steorra/tmp/tmp'
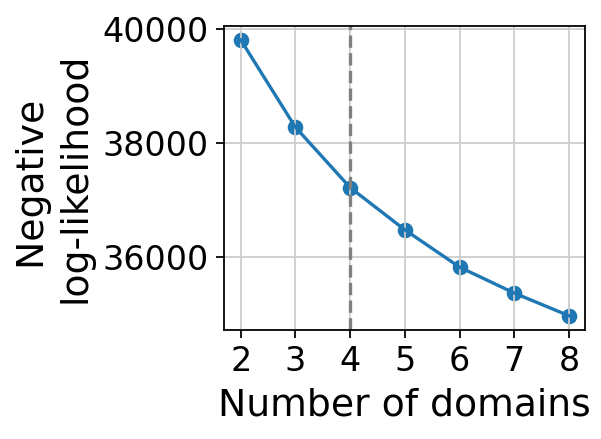
gaston_model, A, S=gas_obj.get_best_model(out_dir=out_dir,
max_domain_num=8,start_from=2)
Calculate the IsoDepth and Labels¶
We can chose the number of layers from IsoDepth
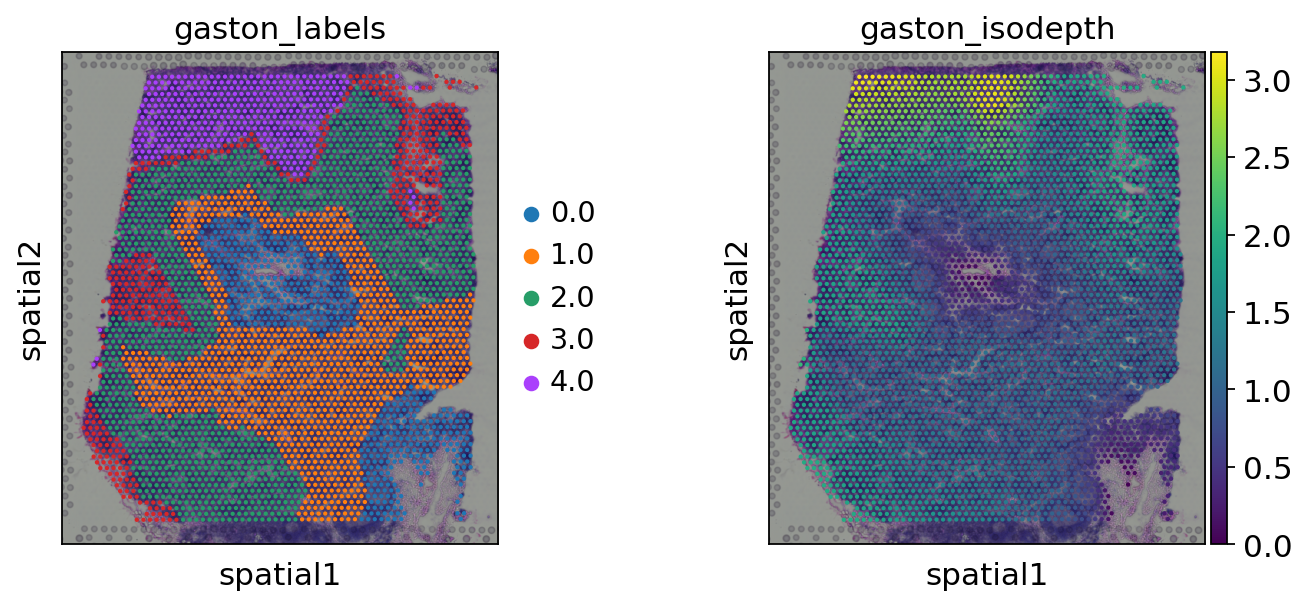
gaston_isodepth, gaston_labels=gas_obj.cal_iso_depth(5)
adata.obs['gaston_labels']=[str(i) for i in gaston_labels]
adata.obs['gaston_isodepth']=gaston_isodepth
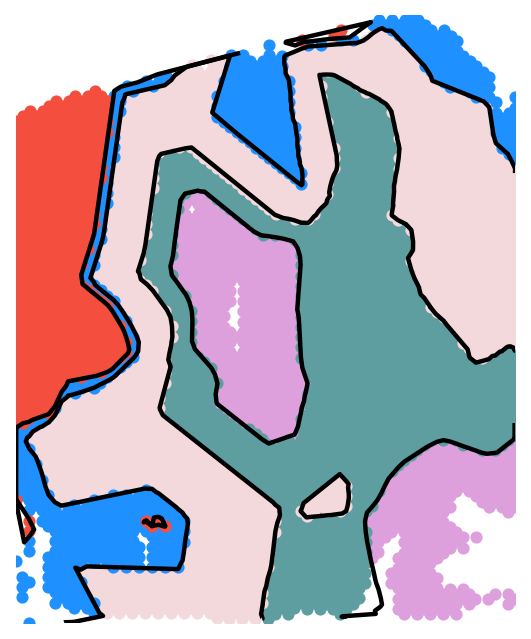
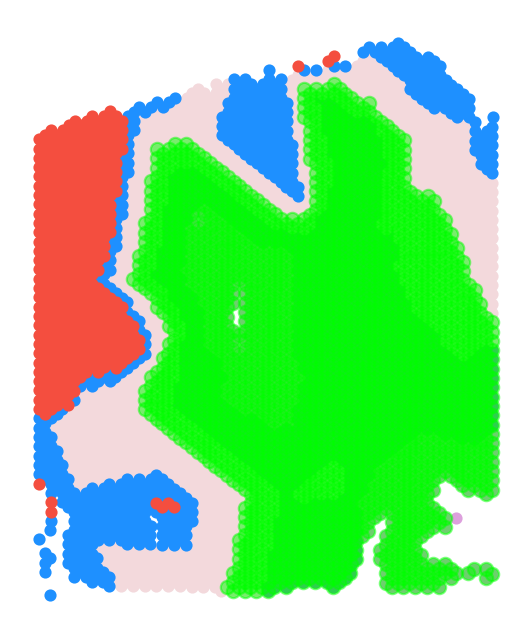
sc.pl.spatial(adata,
color=['gaston_labels','gaston_isodepth'],
show=False)
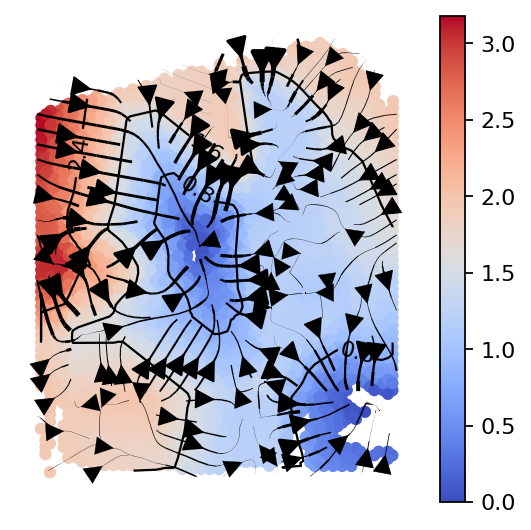
gas_obj.plot_isodepth(show_streamlines=True,
rotate_angle=-90,arrowsize=2,
figsize=(4,4),n_neighbors=100)
Specific to this analysis: restrict to domains (0,1)¶
To isolate the tumor section, we restrict to spots with isodepth lying in a given range. The range of isodepth values will need to be tuned depending on the specific application.
In some cases, the tissue geometry cannot be represented with a single isodepth. In this case, we recommend first subsetting your tissue to the specific region of interest (eg from ScanPy clustering), and then running GASTON
gas_obj.plot_clusters_restrict(
domain_colors,
isodepth_min=0,
isodepth_max=1.5,
rotate_angle=-90,
s=20,lgd=False,
figsize=(4,5)
)
counts_mat_restrict, coords_mat_restrict, gaston_isodepth_restrict, gaston_labels_restrict, S_restrict=gas_obj.restrict_spot(
isodepth_min=0,
isodepth_max=1.5,
adjust_physical=True,
scale_factor=100,
plotisodepth=True,
show_streamlines=True,
rotate_angle=-90,
arrowsize=1, figsize=(4,5),
neg_gradient=True,
n_neighbors=500
)
gas_obj.filter_genes(
umi_thresh = 1000,
exclude_prefix=['MT-', 'RPL', 'RPS']
)
pw_fit_dict=gas_obj.pw_linear_fit()
binning_output=gas_obj.bin_data(
num_bins=15,
q_discont=0.95,
q_cont=0.8
)
The binning_output, cont_genes_layer and discont_genes_layer stored in gas_obj
ad=gas_obj.get_restricted_adata(gas_obj,offset=10**6)
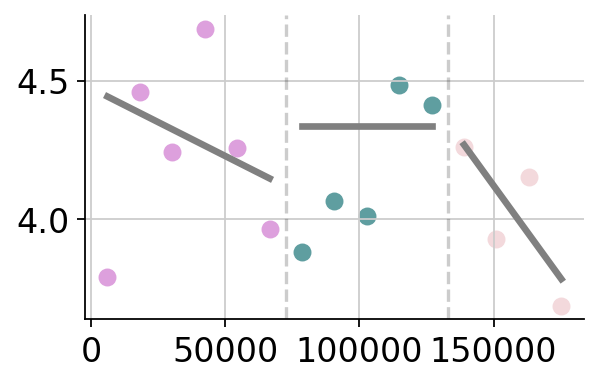
Plot discontinuous and continuous genes¶
We can found the discontinuous genes using gas_obj.discont_genes_layer and found continuous genes using gas_obj.cont_genes_layer
dict(gas_obj.cont_genes_layer)['MARCO']
[0, 2]
Plot cell type vs isodepth (if cell type info available)¶
Load cell type label per spot (from tangram). The tutorial and result could be found in https://omicverse.readthedocs.io/en/latest/Tutorials-space/t_mapping/
We store as N x C dataframe M where M[i,c]=1 if spot i is cell type c, and 0 if not
cell_type_df=ov.pd.read_csv('data/V1_Human_Lymph_Node_celltype.csv', index_col=0)
cell_type_df.head()
| Macrophages_M1 | B_IFN | B_GC_LZ | Mast | B_activated | B_GC_DZ | B_mem | T_CD8+_naive | FDC | B_GC_prePB | ... | Endo | T_CD8+_cytotoxic | T_CD4+ | B_Cycling | T_CD4+_naive | ILC | T_CD4+_TfH | T_CD8+_CD161+ | NKT | NK | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AAACAAGTATCTCCCA-1 | 0.082171 | 0.000017 | 0.021260 | 0.000019 | 0.259048 | 0.030650 | 0.504739 | 0.411314 | 0.000519 | 0.000018 | ... | 0.376284 | 0.043815 | 0.127287 | 0.233432 | 0.006444 | 0.064583 | 0.354094 | 0.170322 | 0.022457 | 0.199254 |
| AAACAATCTACTAGCA-1 | 0.105147 | 0.000004 | 0.087364 | 0.000134 | 0.009732 | 0.005403 | 0.205373 | 0.039582 | 0.110013 | 0.000051 | ... | 0.149988 | 0.154454 | 0.160099 | 0.229329 | 0.703887 | 0.057109 | 0.412450 | 0.003166 | 0.053408 | 0.010387 |
| AAACACCAATAACTGC-1 | 0.112886 | 0.000004 | 0.023793 | 0.000220 | 0.228582 | 0.001881 | 0.536273 | 0.156701 | 0.001156 | 0.000022 | ... | 0.326906 | 0.183677 | 0.110817 | 0.252126 | 0.018042 | 0.083709 | 0.134888 | 0.281477 | 0.001987 | 0.273480 |
| AAACAGAGCGACTCCT-1 | 0.150097 | 0.920552 | 0.083713 | 0.089123 | 0.002341 | 0.031755 | 0.321248 | 0.003961 | 0.169535 | 0.000032 | ... | 0.176462 | 0.398296 | 0.003631 | 0.270192 | 0.353399 | 0.026902 | 0.025387 | 0.015009 | 0.023835 | 0.170472 |
| AAACAGCTTTCAGAAG-1 | 0.281063 | 0.000003 | 0.103263 | 0.000018 | 0.005834 | 0.055721 | 0.433569 | 0.025195 | 0.104654 | 0.000014 | ... | 0.066358 | 0.032739 | 0.019467 | 0.076862 | 0.072655 | 0.002734 | 0.210401 | 0.037577 | 0.003070 | 0.251124 |
5 rows × 34 columns
from omicverse.external.gaston import plot_cell_types
ct_colors=dict(zip(
cell_type_df.columns,
ov.pl.sc_color+ov.utils._plot.palette_28
))
num_bins_per_domain=[10]*len(set(adata.obs['gaston_labels']))
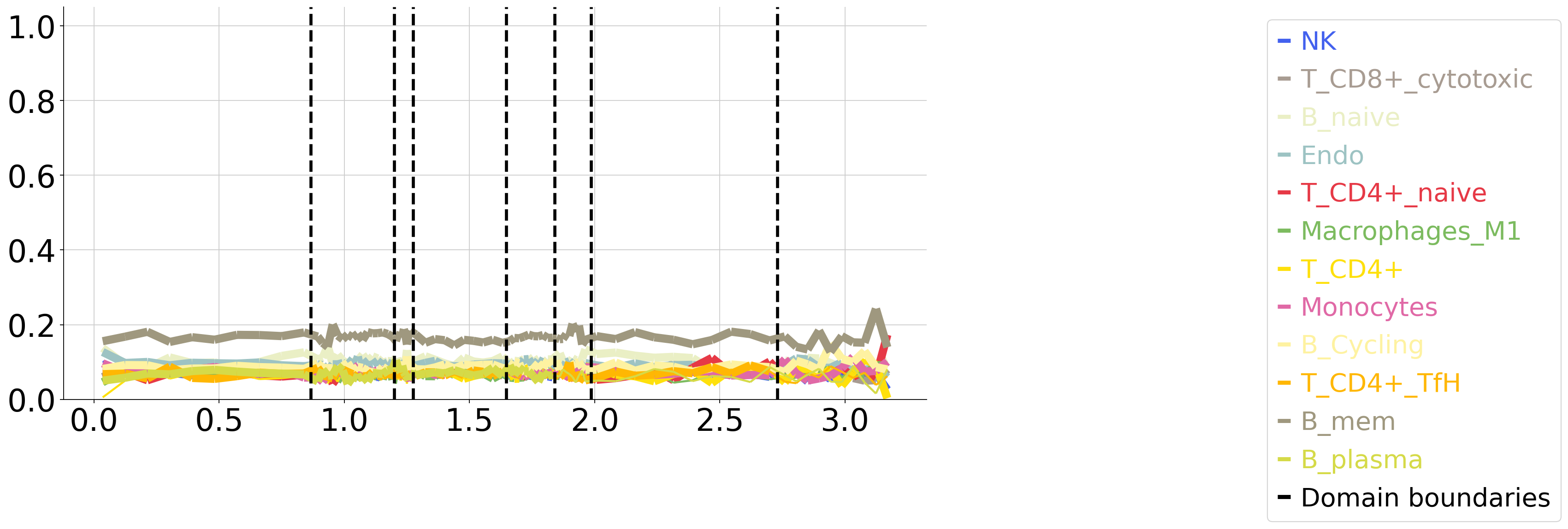
plot_cell_types.plot_ct_props(
cell_type_df,
gaston_labels,
gaston_isodepth,
num_bins_per_domain=num_bins_per_domain,
ct_colors=ct_colors,
ct_pseudocounts={3:1},
include_lgd=True,
figsize=(15,7),
ticksize=30,
width1=8,
width2=2,
domain_ct_threshold=0.5
)